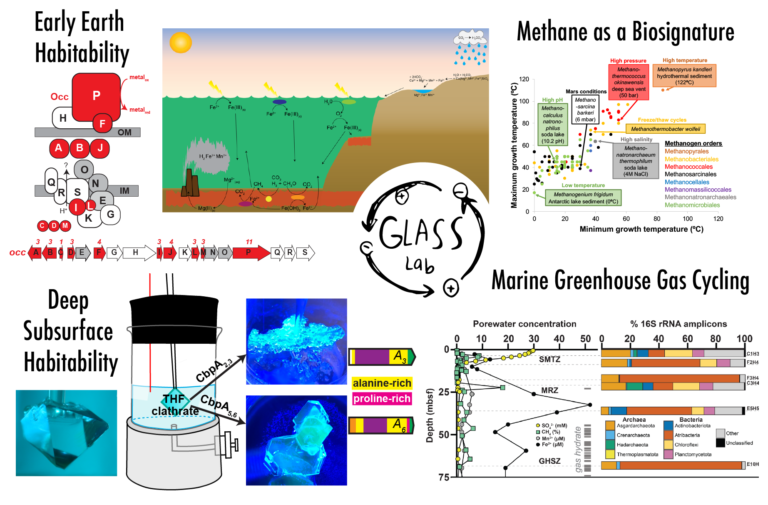
Early Earth Habitability. For the first half of Earth history, the atmosphere was devoid of oxygen, and iron was orders of magnitude more abundant in surface waters than they are today. Solar luminosity was lower than today, and carbon dioxide alone cannot explain the persistence of liquid water in the Archean. Other greenhouse gases, namely methane and nitrous oxide, were likely important for closing the “greenhouse gap” under the Faint Young Sun. With funding from NASA Exobiology, my lab has illuminated how abundant metals on the early Earth sustained microbial production of potent greenhouse gases, and cyanobacterial growth. High methane fluxes were likely sustained by methanogenic archaea in the presence of less reactive iron oxide minerals and low sulfate, as suggested by anoxic enrichments of ferruginous sediments (Bray et al., 2017, Geobiology). Elevated nitrous oxide fluxes could have generated abiotic reactions of ferrous iron and manganese with microbial waste products nitric oxide (Stanton et al., 2018, Geobiology) and hydroxylamine (Cavazos et al., 2018, Marine Chemistry). High manganese may also have helped cyanobacteria to thrive in ferruginous waters (Szeinbaum et al., 2021, Geobiology). While investigating microbial growth on manganese and methane, we discovered a new operon encoding novel cytochrome-rich proteins that is widespread in aquatic betaproteobacteria (Szeinbaum et al., 2020, EM Reports) and developed a new method for co-localizing manganese oxides and microbial cells (Cavazos & Glass, 2020, L&O Methods). Characterization of this new pathway and application of this method to more aquatic environments await further study.
Deep Subsurface Habitability. Methane hydrate clathrates harbor vast reservoirs of frozen carbon on continental margins and permafrost that hold both climatic and economic importance. Methane hydrates are also prevalent throughout other planetary bodies in our solar system. My lab has been investigating the habitability of gas hydrates in the deep subsurface of continental margins and microbial adaptations to these extreme habitats. With C-DEBI funding, we sequenced metagenomes and metaproteomes from 2-70 meters beneath Hydrate Ridge, offshore Oregon. Atribacteria contained diverse mechanisms for survival at low water activity and high salt (Glass et al., 2021, Environmental Microbiology). Mining the Hydrate Ridge metagenomes, we discovered that novel genes encoding proteins with similar amino acid motifs as antifreeze proteins. With NASA Exobiology funding, we took the next step to determine the effect of bacterial proteins on clathrate growth and morphology. In collaboration with GT biochemist Raquel Lieberman, we heterologously expressed these novel proteins, which appear to originate from hydrate-dwelling Chloroflexi bacteria. We experimentally confirmed that these proteins bind to a low-pressure clathrate and change the clathrate’s morphology (Johnson et al., 2020, Crystal Growth & Design). We then engineered an apparatus for high-pressure experiments (Johnson et al., JoVE, 2021). Ongoing investigations are exploring the effectiveness of clathrate-binding proteins as methane hydrate inhibitors.
Methane as a Biosignature. Methane is widely considered to be a key biogenic gas that will be readily detectable in the atmospheres of rocky exoplanets with JWST. The basis for the methane biosignature is that, at Earth’s surface temperatures, no abiotic processes can yield the fluxes of methane generated by anaerobic methanogenic archaea, and this has likely been the case for all of Earth’s history. But these assumptions are for planets with similar redox state and carbon content as Earth, and many potentially habitable rocky exoplanets will be richer in carbon than Earth. A new collaborative project, funded by Scialog/ResCorp, seeks to determine the potential for false-positive methane biosignatures on exoplanets. In collaboration with an atmospheric modeler (Edwin Kite, U Chicago) and an astrophysicist (Smadar Naoz, UCLA), we are studying graphite hydrogenation as a potential source of abiotic methane on rocky exoplanets.
Marine Greenhouse Gas Cycling. Despite significant methane production in continental sediments, the ocean contributes only a small fraction of atmospheric methane emissions thanks to efficient microbial oxidation of methane in the sediments and in the overlying water column. With expanding hypoxic zones in warming oceans, it is essential to know the rates and mechanisms of microbial greenhouse gas cycling that occurs in oxygen minimum zones overlying methane-emitting continental sediments. The microbes that perform this essential ecosystem service remain understudied because they grow slowly and are hard to cultivate. With NSF-OCE funding in collaboration with Frank Stewart (e.g., Glass et al., 2015, Frontiers in Microbiology), we have amassed a large collection of anoxic enrichment cultures from the Eastern Tropical North Pacific Oxygen Minimum Zone. We found that microbial cultures produced another potent greenhouse gas, nitrous oxide, when respiring nitrate anaerobically in the presence of methane; without methane, no nitrous oxide was produced. If the enrichment culture contains organisms that are active in OMZs, it may help explain why the ocean is a minor emitter of global methane emissions yet also a significant source of global nitrous oxide emissions. My lab is currently working to isolate the microorganism responsible for that activity to determine the enzymatic mechanisms underlying this biogeochemical pathway.